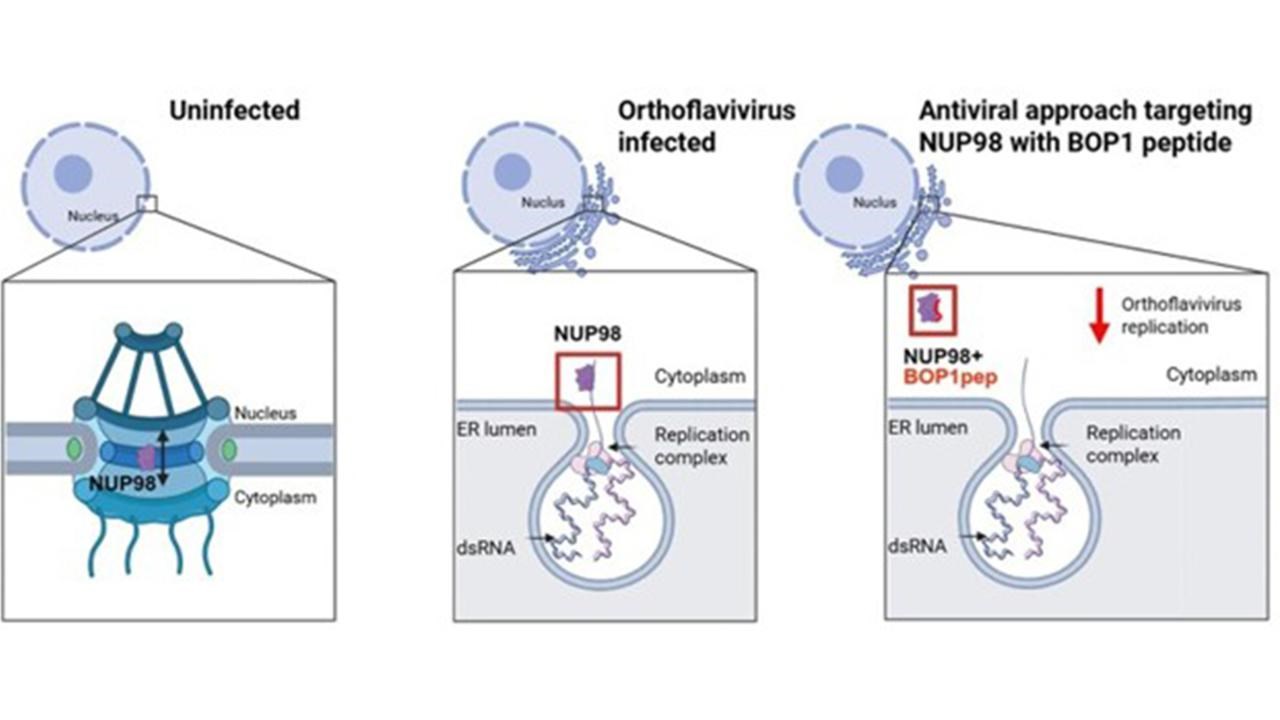
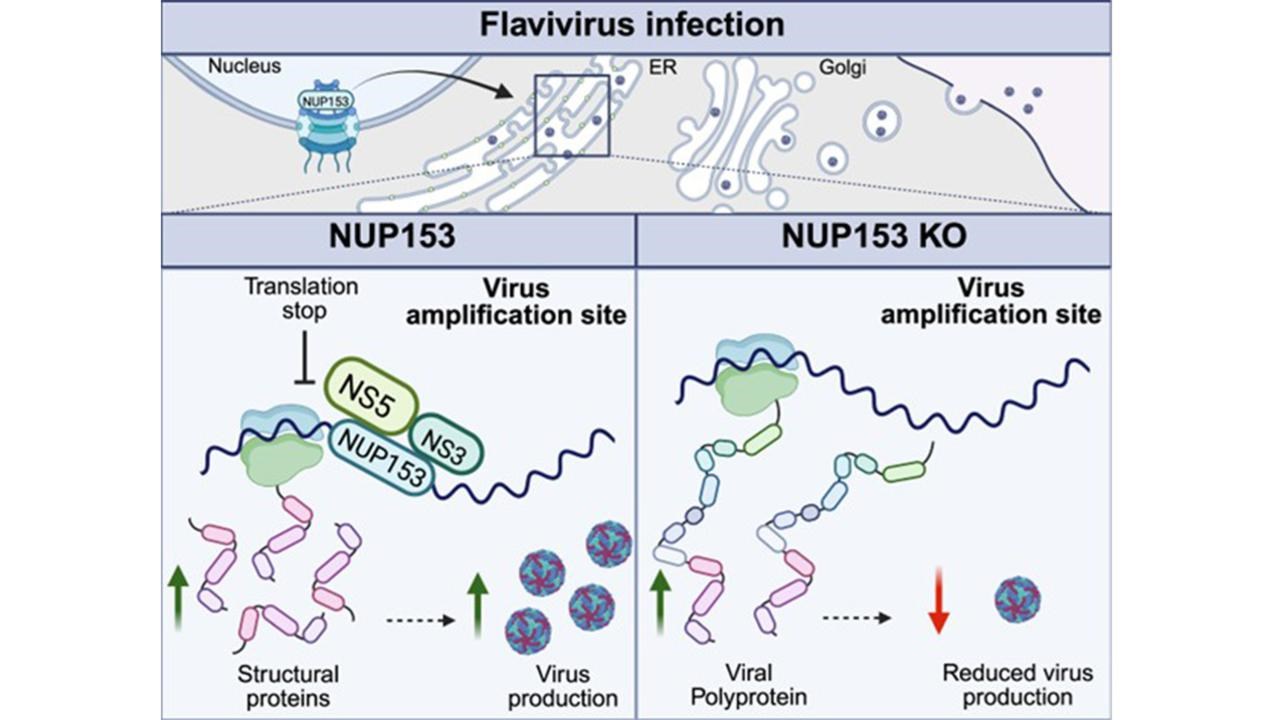
Facts orthoflaviviruses
Orthoflaviviruses are a genus of viruses that include several serious disease-causing pathogens transmitted by mosquitoes and ticks. Millions of people are infected each year, mainly in tropical and temperate regions. These viruses are geographically widespread and are increasing in range due to climate change and global mobility. There are no approved antiviral drugs against most orthoflaviviruses.
Examples of orthoflaviviruses: Tick-borne encephalitis virus (TBEV) – transmitted by ticks and can cause encephalitis. West Nile virus – transmitted by mosquitoes and can lead to neurological disease. Dengue virus – transmitted by mosquitoes and causes dengue fever, which can be life-threatening.